Our organization
Del Corno & Associati is a regulatory and pharmacovigilance consultancy company that provides services to the pharmaceutical industry, aimed at developing and obtaining the necessary authorizations for the marketing of pharmaceutical products, medical devices and food supplements.
Del Corno & Associati started its activity in 2001 in response to an increasing demand for experience in regulatory affairs and business development by pharmaceutical companies and collaborates with a wide range of partners, from large multinationals to small start-up companies up, subcontractors, CROs and medical device manufacturers.
The company is also accredited for product registration with the Italian Medicines Agency.
The company aims to achieve its objectives by working according to 4 criteria: quality, professionalism, excellence and speed.
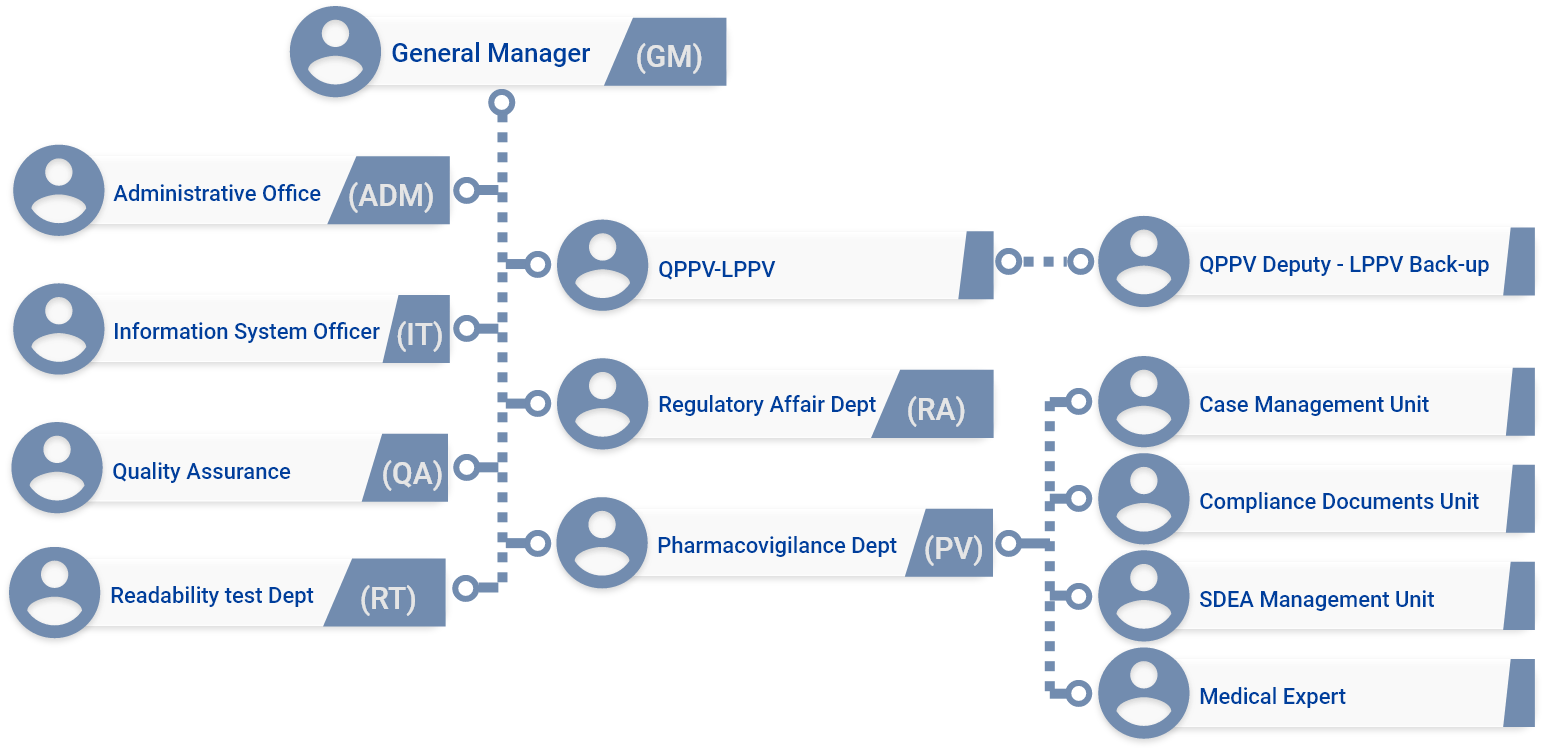
During the activity, a "Quality management system" was implemented that can describe the organizational structure, responsibilities, procedures, methods and resources put in place to prevent any non-compliant situation in all phases for the provision of services.
Del Corno & associati staff, at all levels, constitute the essence of the organization and their full involvement allows the organization to operate successfully.
Outsourcing is an innovative response to the needs of corporate structures that require a resource with professionalism and experience in the field of pharmaceutical regulatory affairs in the short or medium term.
Del Corno & associates provides customized and flexible solutions together with product innovation and quality.
The company employs a highly experienced team, coming from both multinational pharmaceutical companies and the university world.
Del Corno & Associati uses a Quality Management System for its services and continuously improves its effectiveness in accordance with the principles of the UNI EN ISO 9001 standard. The primary objective is the full satisfaction of needs and expectations of Customers in compliance with the regulations in force, with a view to providing the best service while maintaining attention on cost containment, compliance with specifications and delivery times and the utmost competence of the professionals involved. All company functions, external collaborators, and last but not least the suppliers, aware of the company choices, are committed to the continuous improvement of the quality level of the services offered. Del Corno & Associati s.r.l's objective is therefore to: improve the level of services offered to its customers; create a work environment in which everyone is committed to minimizing the possibility of risks and accidents to health and the environment; rationalize and optimize company resources; increase the professional development of their collaborators; establish and consolidate transparent relationships with regulatory and control bodies.
Recognized for investments in "Applied research, technological innovation and other innovative activities for the competitiveness of companies"
20 years from the foundation
Over 120 Italian and foreign customers
Over 2000 PSUR prepared
Prepared over 80 CTD / eCTD dossiers
Hours dedicated to Research & Development projects (over 2800 in 2019).
Drafting of dossiers in CTD format and preparation of electronic dossiers (eCTD)
Management of national and European registration and maintenance procedures (in particular DCP and MRP).
Identification of the most appropriate (efficient and effective) national and international registration strategy for the product, both for complete development and for projects aimed at changes and renewals, planning the times and costs of registration.
Support for the preparation of ASMF and Essential and non-essential variations in manufacturing site
Implementation and management of a tailor made pharmacovigilance system in accordance with European legislation and according to quality system, from the management of reports to the implementation of risk minimization measures
Preparation of PSUR / DSUR, RMP, Addendum Report to module 2.5 and associated reports on request.
Consultancy for the compilation of technical files and their updating, the insertion of the National Database and Directory, authorization of Public Health Advertising at the Ministry of Health and device surveillance service
Continuous education in pharmacovigilance and regulatory.

Via Padre Reginaldo Giuliani, 10A,
20125 Milan ( Italy )